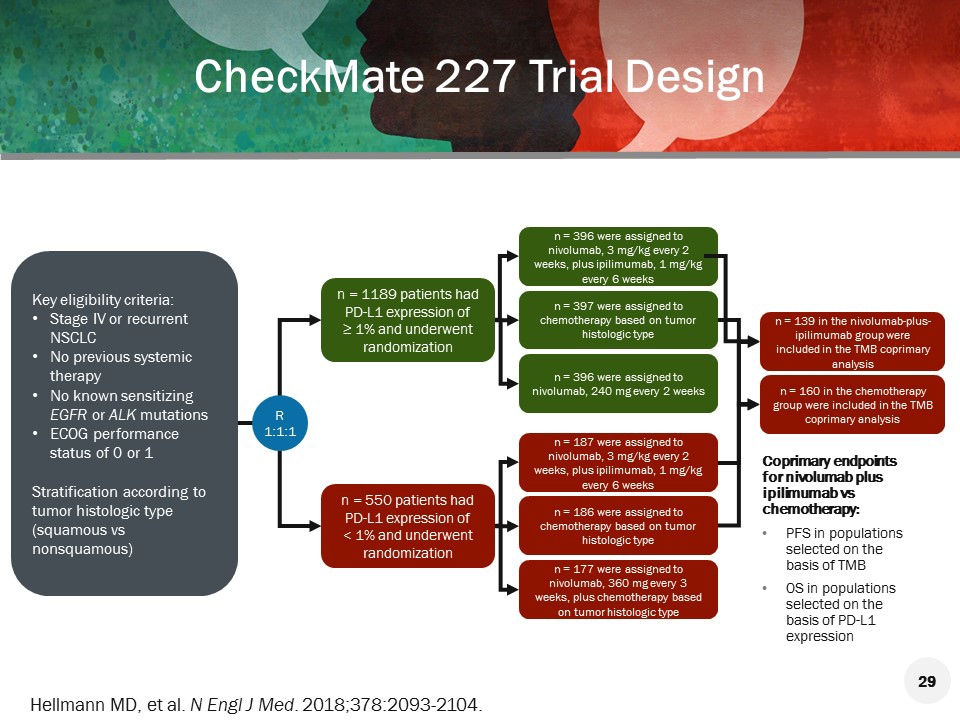
Now it can be said that with this regimen their survival is similar to patients without brain metastases. Trial Design : The purpose of this study is to determine whether Nivolumab, Ipilimumab combined with chemotherapy is more effective than chemotherapy by. He concludes by saying the results from this study can at the very least give hope to patients with brain metastases found at the time of diagnosis, whereas, previously mentioned, they had a terrible prognosis. CheckMate -9LA is an open-label, multi-center, randomized Phase 3 trial evaluating Opdivo (360 mg Q3W) plus Yervoy (1 mg/kg Q6W) combined with chemotherapy (two cycles) compared to chemotherapy alone (up to four cycles followed by optional pemetrexed maintenance therapy if eligible) as a first-line treatment in patients. He says that patients with brain metastases at base-line who were treated with Chemo alone did quite poorly, whereas the patients treated with IPI + NIVO + Chemo did just as well as patients without brain metastases. This study was randomised, so 51 patients were in the NIVO + IPI + Chemo arm, and 50 were in the Chemo only arm. The analysis reported in this study uses patient-level data from the CheckMate 9LA trial and CheckMate 227 Part 1. He begins by recapping on how 30 years ago brain metastases were a truly horrible prognosis with a 6 week median survival after diagnosis.ĭr Carbone then goes onto explain that they identified 101 patients that had brain metastases at base-line. 9501 Background: NIVO + IPI was shown to improve overall survival (OS) and durability of response vs chemo in 1L advanced NSCLC in CheckMate 227 Part 1, regardless of PD-L1 expression. clinical trials and real-world registries. Dr David Carbone talks to ecancer about the results from CheckMate 9LA: First-line nivolumab (NIVO) + ipilimumab (IPI) + chemo in patients with advanced NSCLC and brain metastases.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
